Class 10 Science Sample Paper Term 2 Set A
Please see below Class 10 Science Sample Paper Term 2 Set A with solutions. We have provided Class 10 Science Sample Papers with solutions designed by Science teachers for Class 10 based on the latest examination pattern issued by CBSE. We have provided the following sample paper for Term 2 Class 10 Science with answers. You will be able to understand the type of questions which can come in the upcoming exams.
CBSE Sample Paper for Class 10 Science Term 2 Set A
SECTION – A
1. When different wires of same dimensions but different materials connected such that these are joined end to end then what happens to potential difference across each wire?
Ans. When different wires are connected in end to end then the potential difference across the ends of the combination is distributed across the ends of each one of the wires. Different wires are made up of different materials so they have different resistances. The potential difference across any one of the wire is directly proportional to its resistance.
2. In the network shown in given figure, the ring has zero resistance. Find the resistance between A and B.

Ans. Here, three resistance each of value 6 R are in parallel,

1 / Rp = 1 / 6R + 1 / 6R + 1 / 6R = 1 / 2R
Total resistance between A and B = 2R + R = 3R
3. A fan is rated 20 W; 220 V. Find the current drawn by it, when it is connected to a 220 V supply. Also find its resistance. If the given fan is replaced by a fan of rating 25 W; 220 V, will there be any change in the value of current and resistance? Justify your answer.
Ans. In first case, P = 20 W, V = 220 V

Hence, replacing a 20 W fan by a 25 W fan, across the same source of voltage, the amount of current increases while resistance decreases
OR
State whether an electric heater will consume more electrical energy or less electrical energy per second when the length of its heating element is reduced. Give reasons for your answer.
Ans. We know that, energy, P ∝ 1/R and resistance, R ∝ l (length). So, when length of a heating element is reduced then resistance is also reduced, simultaneously, the energy consumed per second is increased.
4. AB is current carrying conductor in the plane of the paper as shown in given figure. What are the directions of the magnetic field produced by it at points P and Q? Given r1 > r2, where will the strength of the magnetic field be large?
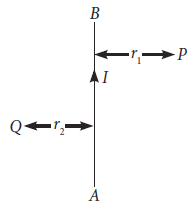
Ans. At point P : The field will be into the plane of paper.
At point Q : The field will be out of the plane of paper
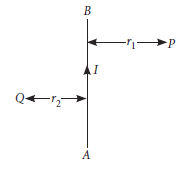
Since point Q is nearer to the conductor than point P (r2 < r1) hence, the strength of the magnetic field at Q is stronger than that at point P.
5. For the coil in the diagram below, when the switch is pressed:
(a) What is the polarity at end A?
(b) Which way will the compass point then?
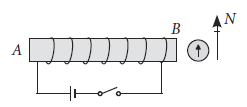
Ans. (a) S-pole
(b) Away from the end B → (Because end B is a N-pole)
6. In Faraday’s experiment if instead of moving the magnet towards the coil we move the coil towards the magnet. Will there be any induced current? Justify your answer. Compare the two cases.
Ans. Yes, there will be an induced current in both the cases as there is a change in the number of magnetic field line associated with the coil or we can say that there is a motion of a magnet with respect to the coil. Same amount of current will be induced and the direction of flow of current will also be the same in the two cases.
OR
A uniform magnetic field is directed in upward direction in the plane of paper. Two charged particles are moving in the field. Find the direction of magnetic force acting on the charged particles.
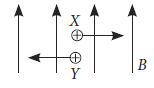
Ans. Use Fleming’s left hand rule,
For X-perpendicular to the plane of paper upwards.
For Y-perpendicular to the plane of paper downwards.
7. Food web consists of number of interconnected food chains through which food energy passes in the ecosystem. Identify the trophic levels (1, 2, 3 and 4) in the figure.
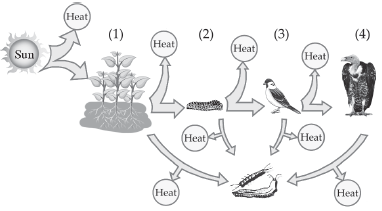
Ans. In the given figure :
1. is T1 – First trophic level i.e., producers.
2. is T2 – Second trophic level i.e., herbivore (Primary consumer).
3. is T3 – Third trophic level i.e., small carnivorous bird (Secondary consumers).
4. is T4 – Fourth trophic level i.e., large carnivorous bird (tertiary consumers).
OR
“Improvements in our lifestyle have resulted in greater amounts of waste generation.” Give one example to support the given statement. Also suggest one change that we can incorporate in our lifestyle in order to reduce non-biodegradable waste.
Ans. The affluent lifestyle has forced people to start using more of disposable items like paper plates, plastic items, polythene, etc. We can reuse polythene bags, plastic containers, etc. to reduce non-biodegradable wastes.
SECTION – B
8. (a) Why is graphite used for making cores of lead pencils?
(b) Why is diamond used for making expensive jewellery?
(c) Name an allotrope of carbon which contains both single and double bonds.
(d) Which allotrope of carbon was discovered by H.W. Kroto, E. Smalley and R.F. Curl?
Ans. (a) Graphite is used for making cores of lead pencils because of its soft, slippery and layered texture.
(b) Diamond is used for making expensive jewelleries because it has a sparkling surface.
(c) Graphite
(d) Fullerene
9. Consider an element 3115X and answer the following questions :
(a) What is its electronic configuration and valency?
(b) To which group and period does it belong?
(c) Is it a metal or a non-metal?
Ans. (a) Atomic number of X is 15. Hence, its electronic configuration is 2, 8, 5.
It has 5 electrons in its valence shell and its valency is 3 (8 – 5 = 3).
(b) It has 3 electron shells, hence it belongs to 3rd period.
It belongs to group-15 as it has five valence electrons.
(c) It is a non-metal because it has more than 3 electrons in its valence shell.
OR
(a) Define Newlands’ law of octaves.
(b) Define Dobereiner’s triads. Give one example of Dobereiner’s triad.
Ans. (a) Newlands’ law of octaves : When elements are arranged in order of increasing atomic masses, the properties of every eighth element is similar to that of the first element.
(b) Dobereiner’s Triads : Groups of three elements which when arranged in increasing order of atomic masses, the atomic mass of the middle element is roughly the average of the atomic masses of other two elements.
Example of triad include : Lithium, sodium and potassium.
10. Asexual reproduction involves a single parent to produce new offsprings. Describe asexual reproduction in Amoeba.
Ans. In Amoeba, asexual reproduction occurs by binary fission. During this, the nucleus divides into two equal halves. The nuclear division is followed by the appearance of a constriction in the cell membrane. The membrane then grows transversely inwards (i.e., centripetally) from near the middle of dividing cell which separates the cytoplasm into two equal parts, each with one nucleus. The two daughter cells separate and behave as two independent organisms.
11. Fertilisation is possible if copulation has taken place during the middle of the menstrual cycle. Give reason. Name the hormone increased during the middle of cycle.
Ans. Middle of the menstrual cycle is the time of ovulation when ovum is released from the ovary and passes into fallopian tube. Thus, chances of fertilisation are more if copulation has taken place during ovulation period. Lutienising hormone (LH) increases during the middle of menstrual cycle.
12. What are variations? How does the creation of variations in a species promote survival?
Ans. The differences existing among the individuals of a species and also among the offspring of the same parents are called variations. Variations are produced to enable the organisms to lead for better adaptation to their environment. An organism adapted better to its environment will have better chances of survival.
OR
(a) How can we say that gametes maintain their purity during reproduction?
(b) State law of independent assortment.
Ans. (a) F1 hybrids when selfed, produced plants with both dominant and recessive phenotypes. It showed that the two unit factors of a character which remain together in an individual do not get mixed up, nor get contaminated and keep their distinct identity. They separate or segregate during gamete formation.
(b) According to the principle of independent assortment, the unit factors of each character segregate independent of one another during gamete formation.
13. Solid waste management is defined as the discipline associated with control of generation, storage, collection, transport and disposal of solid waste materials in an environment friendly and human health friendly way. What are the major regulations of Ecological Solid Waste Management Act 2000?
Ans. Major regulations of Ecological Solid Waste Management Act 2000 (Republic Act 9003) are:
(i) It provides for the implementation of a systematic, comprehensive and ecologically sound management of solid wastes.
(ii) Mandatory segregation of solid wastes at the source such as household, institutional, industrial, commercial and agricultural sources.
(iii) Prohibition on non-environmentally acceptable products and packaging.
(iv) Establishment of materials recovery facility in every locality.
(v) Prohibition against the use of open dumps.
SECTION – C
This section has 02 case-based questions (14 and 15). Each case is followed by 03 sub-questions (a, b and c). Parts a and b are compulsory. However, an internal choice has been provided in part c.
14. Read the passage given below and answer the question that follow :
A series of organic compounds having same functional group, with similar or almost identical chemical characteristics in which all the members can be represented by the same general formula and the two consecutive members of the series differ by —CH2 group or 14 mass unit in their molecular formulae is called a homologous series. For example, all the members of alcohol family can be represented by the general formula, CnH2n+1OH where, n may have the values 1, 2, 3, … etc. The various members of a particular homologous series are called homologues. The physical properties such as density, melting point, boiling point, solubility, etc. of the members of a homologous series show almost regular variation in ascending or descending the series.
(a) What is the general formula of all the members of homologous series of alkynes?
(b) How does physical properties of member of a homologous series changes with increase in molar mass?
(c) The table shows the formulae of three organic compounds that belong to the same homologous series.

What is the general formula of this series? What is the formula of the fourth member of this series?
Ans. (a) Alkynes have the general formula CnH2n-2. e.g., Ethyne (C2H2), Propyne (C3H4), Butyne (C4H6).
(b) Physical properties of members of a homologous series generally increase as the molar mass increases.
(c) Molecular formula of first member : C2H6O
Molecular formula of second member : C3H8O
Molecular formula of third member : C4H10O
Thus, the general formula of the homologous series is CnH2n+2O. The molecular formula of the fourth member of this homologous series is C5H12O.
OR
Draw a table containing first three members of the homologous series of alcohol with their condensed structure and molecular formula.
Ans.

15. Given figure represents longitudinal section of a flower showing the growth of pollen tube.
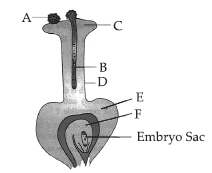
(a) Label the parts `A’, `B’, `C’, `D’, `E’ and `F’ in the following figure.
(b) What are the components of a mature embryo sac?
(c) Assign the role played by `B’ and `F’.
Ans. (a) In the given figure,
A = Pollen grain
B = Pollen tube
C = Stigma
D = Style
E = Ovary
F = Ovule.
(b) The mature embryo sac consists of an egg apparatus (one haploid egg and two synergids), two polar nuclei and three antipodal cells. It is 7 celled and 8 nucleate structure.
(c) Role of B : Pollen tube acts as carrier of male gametes to the embryo sac.
Role of F : Ovule becomes seed after fertilisation.
OR
Discuss briefly about labelled part ‘A’.
Ans. Pollen grain (A) is a haploid, unicellular body with a single nucleus. The outer surface of pollen grain (microspore) may have spines, ridges or furrows called exine and inner layer is intine. At certain places, exine remains thin, and these thin areas are called germ pores.

